For Now,Sulfur Dioxide Pollution becoming seriously,The energy consumption structure dominated by coal is the most important reason for the increasing sulfur dioxide pollution in China.
Desulfurization is to remove SO2 in flue gas , denitration is mainly remove NOx in flue gas (nitrogen), the two substances effluent to the air can transform into acid rain. acid rain is very harmful to the human, The government has been advocating to the environmental protection.Coal - fueled flue gas contains these substances, especially in thermal power plants,now the desulphurization tower has became a necessary requirement for new building power plant.
Sulfur dioxide emissions rate is almost 50% of total emissions capacity, this means the controlling of sulfur dioxide produced in coal combustion is the key to control sulfur dioxide pollution , whichi is the main breakthrough of controlling coal sulfur dioxide pollution .

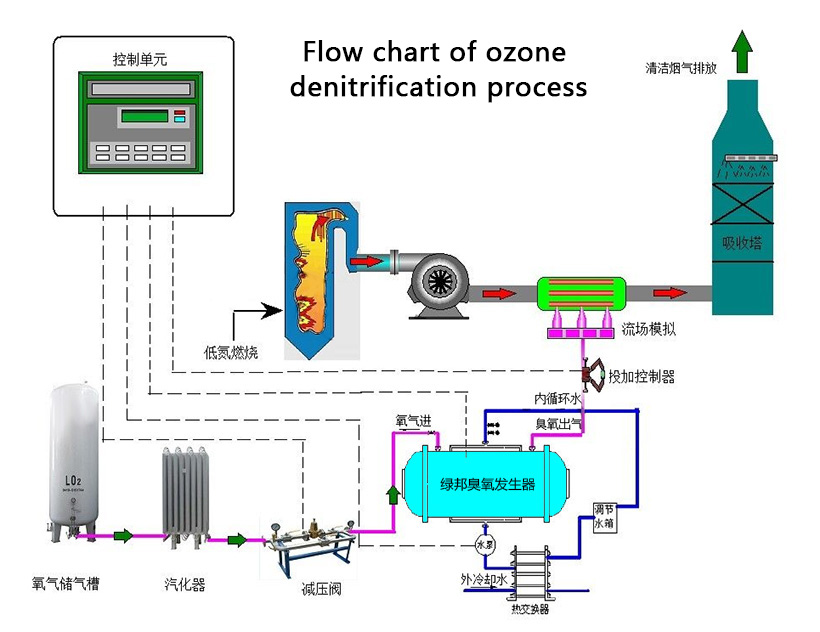
Denitrification of flue gas refers to the reduction of NOX to N2, so as to remove NOX from flue gas.The principle of denitrification of flue gas is to oxidize NO into NO2 by oxidizing it with oxidizer, and then absorb NO2 with water or alkaline solution to achieve denitrification.
Traditional flue gas desulfurization denitration technology can not meet with the requirement of the strict emissions reduction, which is costy and occupy large area, so the development of simple and effective process make a difference.Ozone is used to transform flue gas NOX in N2, the main component of NOX is NO,ozone has its advanced oxidation which can achieve removal effect, and other harmful gas in flue gas also can be removed .As a strong oxidizer, ozone can easily oxidize NO into NO2, N2O3, N2O5 and other high-priced nitrogen oxides that can be dissolved in water to form HNO2 and HNO3.Then Na2S and NaOH solution were adopted for absorption, and NOx was eventually converted to N2 for the purpose of removal. The removal rate of NOx can up to 95% .
Ozone is close to fluorine’s strong oxidation, and it can oxidizing NO into a highly-charged state, so as to improve the water solubility of nitrogen oxides in flue gas.It mainly includes the following reactions:
NO+O3→NO2+O2 (1)Further absorption is required after NO is oxidized by ozone into highly-charged nitrogen oxides.The common absorption solutions are Ca(OH)2, NaOH and other alkaline solutions.The removal effect will be different based on different absorption additions. for example,When using water to absorb the exhaust gas, the NO removal efficiency can reach 86.27%, which is using the solubility of gas in water absorption, there is also other test of using absorbing liquid reduce highly charged nox into N2,then effluent to the air directly.
Name:100kg/h large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:80kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:50kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:40kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:30kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:20kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Name:10kg large ozone generator
Application:De-SOx and De-NOx
Feed Gas Source:Oxygen/Air
Add:13788# Century Road,Jinan City,Shandong Province,China.
Tel:+86-531-88821018
Fax:+86-0531-88825540
Mail:tra@lbozone.com
De-SOx and De-NOxDrinking water treatmentWaste water treatmentAdvanced OxidationOzone Disinfection
Shandong NIPPON Photoelectricity Equipment Co.,LTD
Copyright © 2026 Lbozone All Rights